Dichloromethane (DCM), also known as methylene chloride, has been a favored solvent choice for chromatography due to its ability to solvate heterocyclic compounds, low boiling point, and unique selectivity. Unfortunately, DCM has high neurotoxicity and is a carcinogen. One suggested replacementi is 3:1 ethyl acetate: ethanol. Although this mixture can be used to purify compounds, it isn’t a drop-in replacement for DCM as many people believe but is a starting point to create new methods without DCM. The reason 3:1 ethyl acetate: ethanol behaves differently is apparent in the context of Snyder’s connectivity triangle, where DCM is in group 5, by itself.
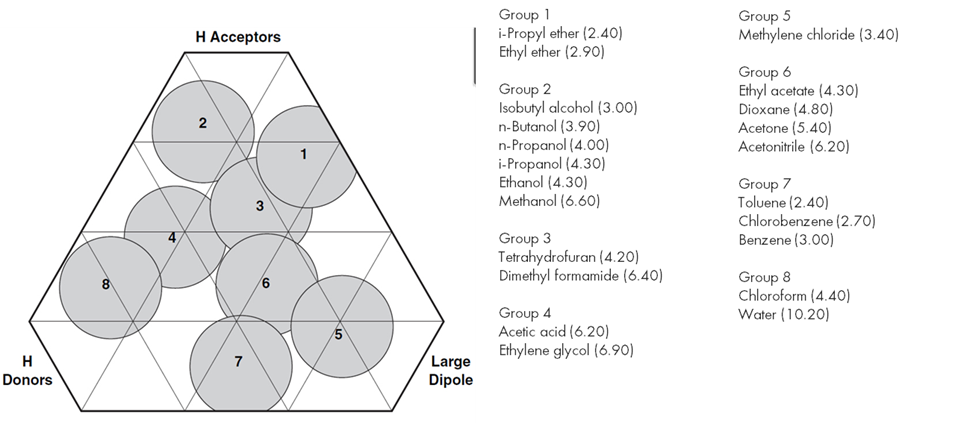
Image: Snyder Selectivity Triangle
Normal phase chromatography commonly uses hydrocarbons as the weak solvent; dichloromethane is one choice for the strong solvent ii,iii. In this case, the hydrocarbon, which may be hexanes, heptane, petroleum ether, or cyclohexane, plays little role in solvent selectivity. Another common solvent system uses DCM as the weak solvent, and methanol as the strong solvent. In this case, both the weak and strong solvents contribute to the selectivity of eluted compounds. An example is shown in the image below:

Image: Dye mixture run with various solvents on thin layer chromatography plates
Left: 20% dichloromethane: 80% methanol
Middle: 50% (3:1 ethyl acetate: ethanol): 50% MeOH
Right: 50% ethyl acetate 50% methanol
The left-most plate, run with DCM, shows a different elution order for the dyes than the plate run with the DCM substitute in the center. The DCM is replaced by ethyl acetate as the weak solvent; the strong solvent is now a mixture of ethanol and methanol. The right-most plate simply uses a mixture of ethyl acetate and methanol. As methanol and ethanol are in the same selectivity group, the substitution of ethanol for a larger amount of methanol doesn't cause the elution order to change, as happened by the substitution of DCM by ethyl acetate: ethanol.
Although 3:1 ethyl acetate: ethanol is a commercially available substitute for DCM for chromatography, other potential replacements used with methanol include methyl tert-butyl etheri, dimethyl carbonateiv, cyclopentyl methyl etheriv, and 2-methyl tetrahydrofuraniv. As none of these are in the same selectivity group as dichloromethane, existing purification methods will need modification to use these solvents.
When developing new chromatography methods, you should try greener, less toxic alternatives before using dichloromethane. Existing purification methods can be made greener although they would need to be changed to account for differences in selectivity.
References:
[i] Taygerly, J. P.; Miller, L. M.; Yee, A.; Peterson, E. A. A Convenient Guide to Help Select Replacement Solvents for Dichloromethane in Chromatography. Green Chemistry 2012, 14 (11), 3020.
[ii] Shoyama, K; Mahl, M.; Seifert, S.; Würthner, F. A General Synthetic Route to Polycyclic Aromatic Dicarboximides by Palladium-Catalyzed Annulation Reaction. J. Org. Chem. 2018, 83(10), 5339–5346
[iii] Boness, V.M.H.; de Oliveira, M.S.; Batista, C.S.C.; Almeida, L.S.; Boffo, E.F.; Villarreal, C.F.; Cruz, F.G. Anti-Inflammatory and Antinociceptive Properties of Kielmeyerone A, a Chromenone Isolated from the Roots of Kielmeyera reticulata. J. Nat. Prod. 2021 84 (8), 2157-2164
[iv] MacMillan, D.S.; Murray, J.; Sneddon, H.F.; Craig Jamieson, C.; Watson, A.J.B. Replacement of dichloromethane within chromatographic purification: a guide to alternative solvents. Green Chem., 2012, 14, 3016-3019
About Teledyne ISCO - Chromatography
The Teledyne ISCO Chromatography product line includes instruments and accessories for the purification of organic compounds in normal-phase and reversed-phase separations, and for bio-purification of proteins, peptides, and other biopolymers. For more articles from Teledyne ISCO, please visit our blog.
